Over the past 5 years, Provectus has developed Provect-OX®, Provect-OX2™, Provect-“EBR®” and other ISCO technologies with a focus on improved safety, ease of use, permanence of performance (manages rebound) and expanded implementability (ISCO Permeable Reactive Barriers). These technologies are typically more cost-efficient and offer tangible technical benefits over conventional ISCO approaches.
For example, base activation technologies require that the pH of the aquifer medium exceed 10.5 in order to activate persulfate. Increasing aquifer pH above 10 works as a persulfate activation chemistry. But from a bioremediation perspective (and an overall aquifer “health” perspective) extreme basification as the activation chemistry for persulfate-based ISCO seems counterproductive to a remedial action. There are at least five reasons to critically assess the use of base as the activation chemistry for persulfate-based ISCO:
1. There are very few microorganisms on our planet that can tolerate highly basic (pH >10) environments. These are termed “extremophiles” and no known halorespiring bacterium is among them. Yes, over time (perhaps years) the microbial populations will eventually recover – albeit altered – once the aquifer pH returns to more hospitable conditions. But there are more advanced, safer ways to achieve the desired response of synergistic ISCO and sustained bioremediation (the latter being essential to manage contaminant rebound to avoid the need for repeat treatments).
2. When using NaOH as the basifying agent the two materials need to be mixed on site. This complicates field work and requires careful management of several important health & safety issues associated with material handling and storage. Today there are safer, easier to use ISCO technologies that are will save time and money when utilized. When using lime to activate persulfate, there are well-known issues associated with physical encrustation and binding of contaminants to soil: the contaminants are coated in lime crust and entombed within the soil matrix, hidden from the aqueous samples. Once that crust dissolves, the contaminants rebound because in fact, they were always present as part of the precipitates. NOTE: the same phenomenon has been observed with permanganate (MacKinnon and Thomson, 2002; Urynowicz and Siegrist 2005).
3. When using Ca peroxide to elevate pH to activate persulfate, an espoused benefit is to provide oxygen to support secondary bioremediation of residual contaminants. Oxygen can indeed enhance the aerobic biodegradation of reduced organic compounds such as BTEX and TPH once the pH has lowered to ca. <8. But it will take time (perhaps years) for the aquifer to recover from extreme basification (or acidification) and, unfortunately, the oxygen release feature will only effectively last perhaps 6 to 9 months. Lastly, it is important to note that chlorinated compounds, in general, are not very amenable to aerobic biodegradation hence this chemistry seems poorly fitted for halogenated solvent-impacted sites.
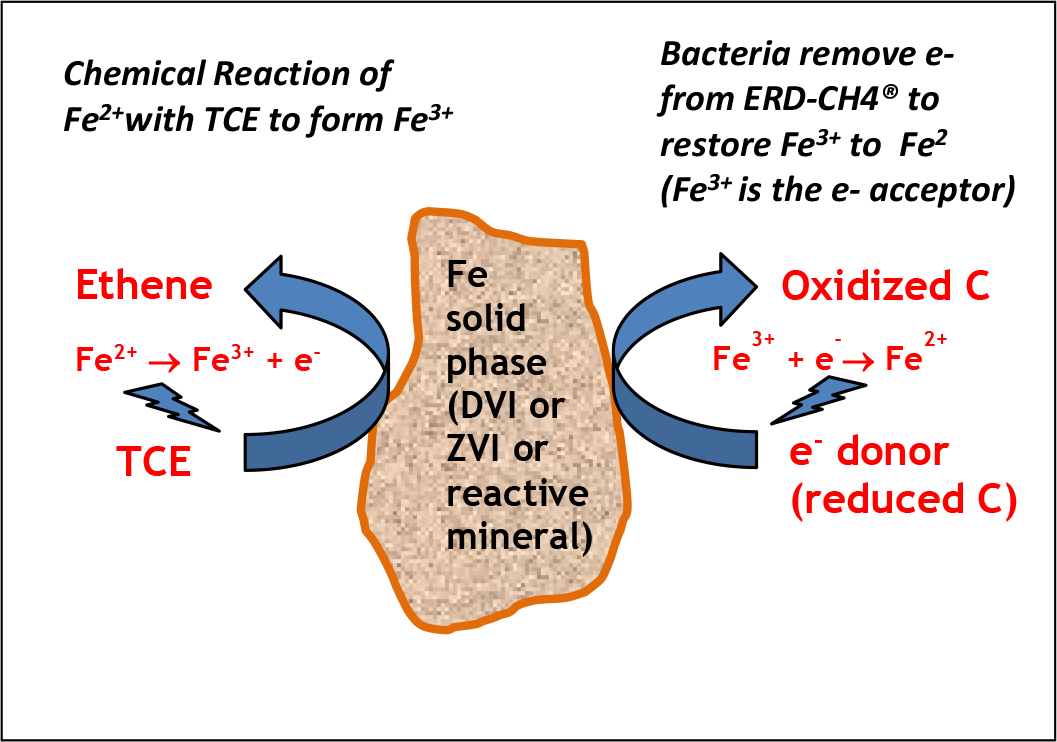
4. For every 1,000 lbs of persulfate applied in the field, there will be 800 lbs of residual sulfate: this may need to be managed to avoid potential complications associated with a newly generated secondary sulfate/hydrogen sulfide plume. Unfortunately, base activation chemistries do nothing to actively manage the residual sulfate. However, in the presence of iron at proper proportions the iron-reducing bacteria work in synergy with the sulfate-reducers to biodegrade many organic compounds. In so doing, they form in situ (for free!) reactive iron-sulfide minerals such as Magnetite and – in the presence of sulfur source – reactive iron sulfides (e.g., Mackinawite) to yield abiotic reductive dechlorination. Critically, these materials are formed in place, where they are needed (as compared to when extraneous sources of Mackinawite or related materials are injected and they are only present where they were physically emplaced). These abiotic pathways support further dechlorination (Chen et al., 2014; Lovely and Phillips, 1988; Weber et al., 2006) and can persist for many years (if an electron donor is available) being catalyzed by indigenous bacteria. Notably, biotransformation of Fe2+ does not require direct contact with the iron solid phase as a variety of naturally-occurring biological molecules, such as humic acids, can facilitate electron shuttle dynamics.
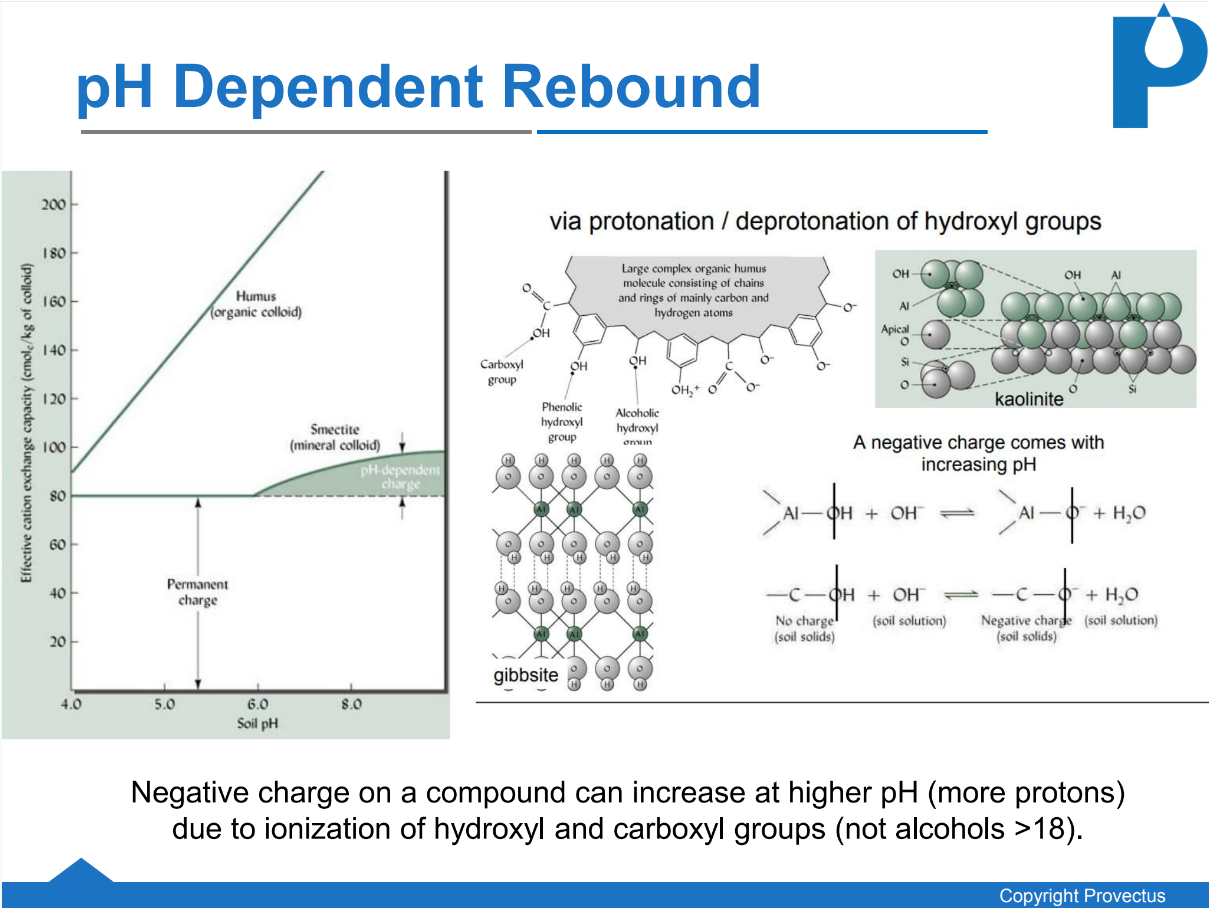
5. Lastly, it has been fully established that all organic compounds will obtain a negative charge at elevated pH (see below). As such, at pH 10 compounds with a hydroxyl or carboxyl functional group will have a negative charge and may not be detected by conventional analytical methods such as US EPA 3545; this is especially true with lime-activation (FMC [now PeroxyChem], 2010). Groundwater samples are commonly preserved via acidification, but soil samples are commonly stored on ice. Therefore, it is especially important to ensure that soil samples collected for chemical analyses are adjusted to pH 7 prior to extraction otherwise analytical procedures may not detect charged/encrusted contaminants leading to false data and inaccurate assessment of ISCO performance.

Celebrating a Solution
The use of a pre-mixed, self-activating, inherently buffered ISCO amendments addresses these issues (Click here for a Comparative Analysis). In 2014, Provectus debuted Provect-OX® as the industry’s first self-activated, persulfate-based ISCO amendment specifically designed to support sustained bioremediation processes and therefore minimize rebound (US patent 9,126,245; patents pending). In 2017, we improved the amendment by adding Terr-OR™ as a buffer and stabilizer for reactive ferrate (patents pending). In 2018, we announced Provect-OX2™ as a combination of sodium and potassium persulfate plus Terr-OR® buffer/ferrate stabilizer to allow for extended in situ chemical oxidation (ISCO) uniquely coupled with long-term (> 3 years) enhanced bioremediation. How? Ferric iron (Fe III) is used as a safe and effective means of activating the persulfates. Subsequently, iron and sulfate serve as alternative electron acceptors to drive biodegradation, and myriad iron-sulfate biogeochemical reactions geologically form reactive minerals in situ to help manage contaminant rebound, long-term.
The synergistic chemistries render Provect-OX® and Provect-OX2™
“Much Greater Than One”